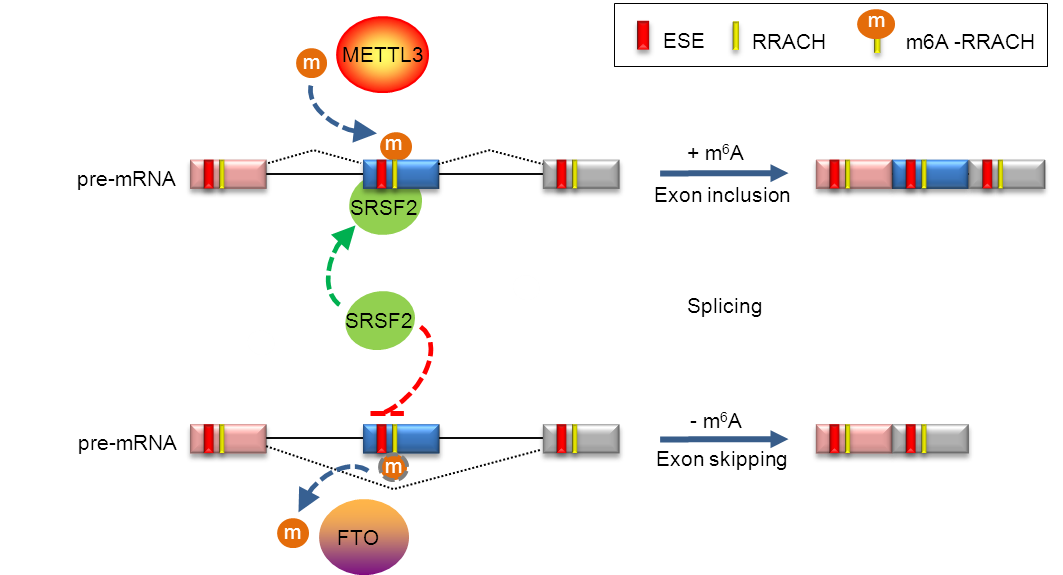
Since first identified in mammalian mRNA in the 1970s, N6-methyladenosine (m6A) has been proposed to regulate mRNA processing including alternative splicing, RNA degradation and translation. In both mammals and yeast, m6A methylation of RNA preferentially occurs within the consensus sequence RRACH (R=G or A; H=A, C or U) in gene coding regions and 3’UTRs, implicating fundamental roles in RNA processing and translational control. The recent identification and characterization of the m6A methyltransferase WMM complex (WTAP-METTL3-METTL14), the demethylases FTO and ALKBH5, and YTH-domain containing binding proteins highlighted the biological significance of m6A methylation. However,the role of m6A in mRNA processing remains largely unknown. The lab of Prof. YANG Yungui from Beijing Institute of Genomics, Chinese Academy of Sciences (CAS), revealed that FTO-dependent demethylation of N6-methyladenosine regulates mRNA splicing and is required for adipogenesis. The work has been published online in Cell Research. The study shows that FTO expression and m6A levels are inversely correlated during adipogenesis. FTO depletion blocks differentiation and only catalytically active FTO restores adipogenesis. Transcriptome analyses in combination with m6A-Seq revealed that gene expression and mRNA splicing of grouped genes are regulated by FTO. M6A is enriched in exonic regions flanking 5’- and 3’-splice sites, spatially overlapping with mRNA splicing regulatory SR protein exonic splicing enhancer binding regions. Enhanced levels of m6A in response to FTO depletion promotes the RNA binding ability of SRSF2 protein, leading to increased inclusion of target exons. FTO controls exonic splicing of adipogenic factor RUNX1T1 by regulating the m6A level around splice sites and thereby modulate differentiation. These findings provide compelling evidence that FTO-dependent m6A methylation may function as a novel cis-regulatory element of RNA processing and play a critical role in the regulation of adipogenesis. This research is supported by CAS, Ministry of Science and Technology and Natural Science Foundation of China. Cooperative role of m6A in regulating SRSF2 protein at spliced exons (Image by YANG Yungui's lab).