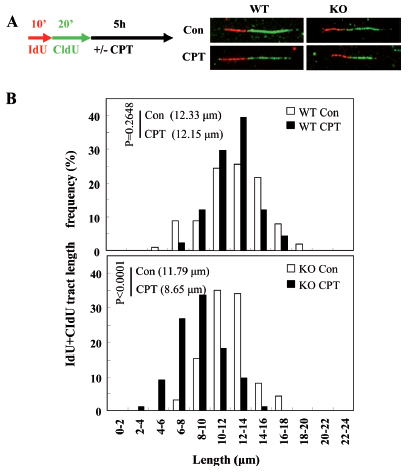
REV1 is a eukaryotic member of the Y-family of DNA polymerases. Compelling studies reveal that REV1 is involved in translesion DNA synthesis and genome mutagenesis. It has been shown that reducing the level of REV1 not only sensitizes tumor cells to chemotherapy, but also diminishes the chance for tumor cells to develop drug resistance. Recently, some studies reported that REV1 is also involved in homologous recombination (HR), which is an important repair mode for DNA double-strand breaks (DSBs), in budding yeast, chicken DT40 cells, Drosophila melanogaster and human cells. However, it remains unclear how REV1 is recruited to sites where HR is processed. The groups led by Drs. GUO Caixia from Beijing Institute of Genomics and TANG Tieshan from Institute of Zoology, CAS, revealed for the first time that FANCD2 and REV1 cooperate in the protection of nascent DNA strands from nuclease degradation in response to replication stress. The article has been published online in Nucleic Acids Research. Using HR reporter cells, this work not only confirms that REV1 is directly involved in regular HR, but also shows that REV1 is required for replication-associated HR. Then, by employing laser microirradiation technique, REV1 is found to be accumulated to DSBs in a manner dependent on its ubiquitin-binding motifs, on RAD18, and on monoubiquitinated FANCD2 (FANCD2-mUb). Expression of a FANCD2-Ub chimeric protein in RAD18-depleted cells could enhance REV1 assembly at laser-damaged sites, suggesting that FANCD2-mUb functions downstream of RAD18 to recruit REV1 to DNA breaks. Additionally, REV1 enrichment at DNA damage stripes also partially depends on BRCA1 and BRCA2. Intriguingly, REV1 is found to play a role in protection of nascent DNA strands from nuclease degradation after camptothecin or hydroxyurea treatment by DNA fiber assay. This study reveals that REV1 plays multiple roles in DNA damage response, which deepens our knowledge of REV1 as a potential drug target in cancer therapy. The work is supported by CAS, Ministry of Science and National Natural Science Foundation of China. The collaborators include Dr. PEI Huadong from Beijing Proteome Research Center, Dr. Benjamin Chen from UT Southwestern Medical Center and the group of Dr. Niles de Wind from Leiden University Medical Center, etc. REV1 protects nascent DNA strands from nuclease degradation