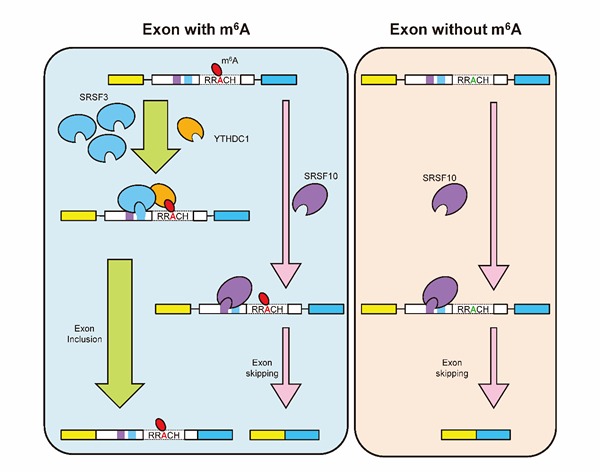
N6-methyl-adenosine (m6A) is one of the most common and abundant modifications on RNA molecules present in eukaryotes. Epitranscriptomic m6A methylation is catalyzed by a multicomponent methytransferase complex composed of at least three subunits: METTL3, METTL14 and WTAP, removed by demethylases FTO and ALKBH5, and recognized by cytoplasmic YTHDF1 and YTHDF2, and modulates cytoplasmic mRNA stability and translation. The regulatory role of m6A and its nuclear binding protein YTHDC1 in the nuclear mRNA processing (i.e. pre-mRNA splicing) remains an enigma. The lab of Prof. YANG Yungui from Key Laboratory of Genomic and Precision Medicine, Collaborative Innovation Center of Genetics and Development, Beijing Institute of Genomics, Chinese Academy of Sciences revealed that the nuclear m6A reader YTHDC1 regulates mRNA splicing, the work has published online in Molecular Cell on February 11, 2016. Their studies identified several SR families of proteins (SRSF1, SRSF3, SRSF7, SRSF9 and SRSF10) as potential YTHDC1 interacting partners by MASS spectrometric analysis of affinity purified SFB-YTHDC1 complex. Scientists used transcriptome assay in combination with PAR-CLIP-Seq assay to reveal that YTHDC1 and SRSF3 promotes inclusion whereas SRSF10 promotes exclusion of their target exons and the spatial association between YTHDC1, SRSF3 and SRSF10. They further demonstrated that YTHDC1 is competitively bound by SRSF3 and SRSF10. Further investigation of the interaction revealed that the interaction takes place between N-terminus of YTHDC1 and C-terminus of SRSF3 and SRSF10. Immunofluorescence assay revealed that nuclear speckle localization of SRSF3 is enhanced whereas that of SRSF10 is reduced by YTHDC1 and the m6A binding ability of YTHDC1 is crucial for this effect as demonstrated by immunofluorescence based rescue assay. Moreover, the RNA binding ability of SRSF3 and SRSF10 is differentially regulated by YTHDC1 via its m6A binding activity. They clearly demonstrated that YTHDC1 and SRSF3 promote inclusion whereas SRSF10 promotes skipping of the target gene by using minigene reporter construct. These observations have established m6A as an epitranscriptomic mark critical for mRNA splicing control through its reader YTHDC1. This research is supported by CAS Strategic Priority Research Programs, Ministry of Science and Technology, Natural Science Foundation of China. m6A reader YTHDC1 modulates interaction of cis- and trans- acting factors and promotes exon inclusion of targeted mRNAs through facilitating SRSF3 while blocking SRSF10 mRNA-binding.(Image by XIAO Wen) Contact: YANG Yungui E-mail: ygyang@big.ac.cn