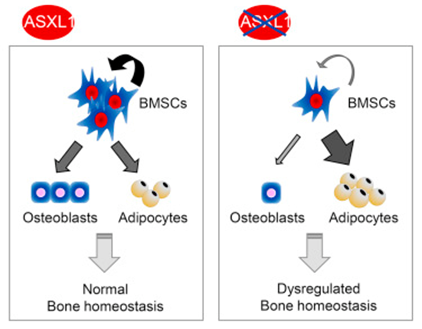
A new research reported that loss of Asxl1 alters self-renewal and cell fate of bone marrow stromal cell, leading to Bohring-Opitz-like syndrome in mice. This study revealed a pivotal role of ASXL1 in the maintenance of BMSC functions and skeletal development. The researchers included Dr. WANG Qianfei from Beijing Institute of Genomics, Chinese Academy of Sciences, and Dr. YANG Fengchun from University of Miami Miller School of Medicine. The study was recently published in the Stem Cell Reports. Bone marrow stromal cells (BMSCs) are multi-potent progenitor cells with self-renewal capabilities and multi-lineage differentiation potentials, including osteogenesis and adipogenesis. The relation between osteogenesis and adipogenesis in BMSCs is critical for normal bone homeostasis. Skewed cell fate of BMSCs can lead to developmental defects. Bohring-Opitz syndrome (BOS) is a heterogeneous genetic disease with severe developmental defects and early childhood mortality. De novo ASXL1 mutations are found in patients with BOS . ASXL1 belongs to the enhancer of trithorax group (TrxG) and polycomb group (PcG), which is required for maintenance of both activation and silencing of Hox genes. However, the cellular and molecular mechanisms by which ASXL1 mutation causes BOS remain unclear. Researchers found that Asxl1 global loss as well as conditional deletion in osteoblasts and their progenitors led to significant bone loss and a markedly decreased number of BMSCs compared with wild-type littermates in Asxl1-targeted murine models. Asxl1−/− BMSCs displayed impaired self-renewal and skewed differentiation, away from osteoblasts and favoring adipocytes. RNA-sequencing and related bioinformatics analyses revealed altered expression of genes involved in cell proliferation, skeletal development, and morphogenesis. Furthermore, gene set enrichment analysis showed decreased expression of stem cell self-renewal gene signature, suggesting a role of Asxl1 in regulating the stemness of BMSCs. Importantly, re-expression of Asxl1 normalized NANOG and OCT4 expression and restored the self-renewal capacity of Asxl1−/− BMSCs. Somatic ASXL1 alterations have also been reported in elderly patients with myeloid malignancies, including myelodysplastic syndrome, chronic myelomonocytic leukemia, and acute myeloid leukemia. This study unveils a pivotal role of ASXL1 in the maintenance of BMSC functions and skeletal development, providing great promise for the future treatment of metabolic bone diseases and cancers. This work was supported in part by grants from the National Natural Science Foundation of China, and the External Cooperation Program of BIC, Chinese Academy of Sciences. ASXL1 plays a pivotal role in the maintenance of BMSC functions and skeletal development (Image by YANG Fengchun's group) Contact: Prof. WANG Qianfei Email: wangqf@big.ac.cn