The maternal-to-zygotic transition (MZT) involving maternal RNA and protein depletion and zygotic genome activation (ZGA) is one of the most important events during early embryogenesis. In zebrafish, several factors have been indicated to be essential in regulating maternal mRNA decay through maternal and zygotic pathways, including the zygotically transcribed microRNA miR-430, suboptimal codon usage, N6-methyladenosine (m6A) and uridylation. However, how the stability of maternally supplied transcripts are maintained during MZT remains largely unknown. 5-Methylcytosine (m5C) is a prevalent mRNA modification. In the previous study, Prof. YANG Yungui’s team at Beijing Institute of Genomics of the Chinese Academy of Sciences (CAS) has employed the novel RNA-BisSeq approaches they developed to map the mRNA m5C profiles in mammalian cells and tissues, and demonstrated that m5C mediates nuclear export of methylated RNAs through its binding protein ALYREF. Another report from this team also illustrated that cytoplasmic m5C binding protein YBX1 regulates mRNA stability and bladder cancer development. A most recent cooperative study led by Prof. YANG Yungui and Prof. LIU Feng from Institute of Zoology of the CAS and Prof. MA Jinbiao from Fudan University has revealed that m5C maintains mRNA stability through the newly identified m5C binding protein Ybx1 during MZT. This work has been published in Molecular Cell on August 6. In this work, they illustrated the first single-base resolution RNA m5C landscape in zebrafish embryos. Based on the high-throughput sequencing analyses, they found that m5C-modified maternal mRNAs display higher stability than non-m5C-modified mRNAs during MZT. Using oligo-pulldown and IP-MS combined with ITC and protein crystallography technologies, they identified the m5C binding protein Ybx1 preferentially recognizes m5C-modified mRNAs through π-π interactions with a key residue, Trp45, in Ybx1’s cold shock domain (CSD). Furthermore, Ybx1 deficiency leads to early gastrulation arrest. Through Co-IP, GST-pulldown and high-throughput sequencing, they found that poly(A)-binding protein Pabpc1a is an interactive partner of Ybx1 in zebrafish embryos. Consistent with the results of Ybx1 deficiency, Pabpc1a depletion led to similar defective phenotypes, and meanwhile, the stability of Ybx1 target mRNAs decreased significantly. Finally, they used single gene reporter system to verify that Ybx1 recruits Pabpc1a to maintain the stability of maternal m5C-modified mRNAs and facilitate zebrafish early embryonic development. This work demonstrated that m5C modification maintains maternal mRNA stability through its binding protein Ybx1 which recruits Pabpc1a and then facilitates maternal-to-zygotic transition. Schematic model of the dynamic regulation of maternal mRNA stability by m5C during zebrafish MZT (Image by YANG Yungui's ) Contact: 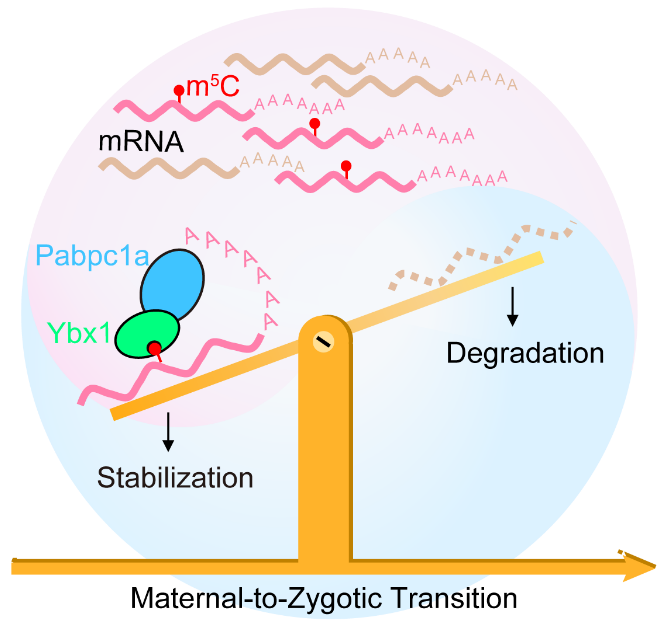
Dr. YANG Yungui
Email: ygyang@big.ac.cn
CAS Key Laboratory of Genomic and Precision Medicine
