PINK1-PRKN/Parkin axis plays a crucial role in mitochondrial quality control through PINK1-dependent phosphorylation of both Parkin and its ubiquitin leading to the fully activated Parkin E3 activity. Several lines of evidence suggest that mitochondrial dysfunction is strongly implicated in the etiology of the neurodegenerative disease. Deubiquitinating enzymes (DUBs) functionally reversing PRKN ubiquitination are critical in controlling the magnitude of PRKN-mediated mitophagy process. However, potential DUBs that directly target PRKN and antagonize its pro-mitophagy effect remains to be identified and characterized. A most recent study led by Prof. ZHAO Yongliang from Beijing Institute of Genomics, Chinese Academy of Sciences (CAS) has revealed that USP33, one of DUB family, deubiquitinates PRKN on depolarized mitochondria and USP33 silencing protects SH-SY5Y human neuroblastoma cells from the neurotoxin MPTP-induced apoptotic cell death. This work has been published in Autophagy on August 26. In this work, they demonstrated firstly that USP33/VDU1 is localized at the outer membrane of mitochondria, and interacts with USP33. Moreover, their interaction is substantially enhanced on depolarized mitochondria. Further cellular and in vitro assays demonstrated that USP33 deubiquitinates PRKN mainly at Lys435, and prefers to remove K6, K11, K48 and K63-linked ubiquitin conjugates from PRKN. It is well established that K48- and K63-linked Ub chains mainly target the proteins for proteasome degradation and diverse signaling pathways, respectively. Intriguingly, USP33 deficiency enhanced both K48- and K63-linked PRKN ubiquitination, but only K63-linked PRKN ubiquitination was significantly increased under mitochondrial depolarization. Further, USP33 knockdown promotes the recruitment of PRKN to depolarized mitochondria leading to an enhanced mitophagy versus a significantly decreased apoptotic cell death induced by a neurotoxin MPTP. The findings reveal a novel regulatory mechanism of deubiquitinase USP33 in controlling the magnitude of PRKN-mediated mitophagy process, and offer the important insights into dynamic balance of ubiquitination and deubiquitination in the maintenance of mitochondrial self-homeostasis. In particular, the findings provide the important basis for the potential biological significance of mitophagy process and a potentially valuable strategy for treatment of the neurodegenerative disease. Model depicting the antagonizing effect of USP33 on PRKN-mediated mitophagy. (Image by ZHAO Yongliang's lab) Contact: Dr. ZHAO Yongliang 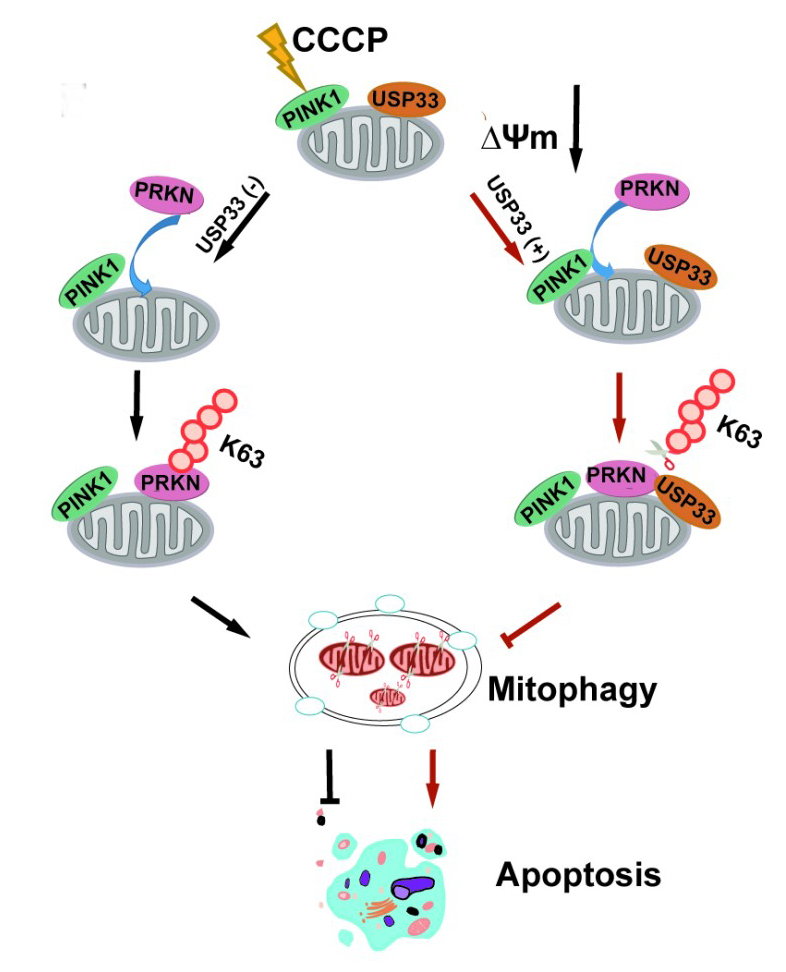
Email: zhaoyongliang@big.ac.cn
Beijing Institute of Genomics
