Researchers Report a New Immune Evasion Mechanism Achieved by FTO-mediated Regulation of Glycolytic Metabolism
CD8+ T cells are the key immune cells to recognize and eliminate tumor cells, but their functions are usually inhibited by the immunosuppressive tumor microenvironment.
Previous studies have shown that tumor cells utilize diverse epigenetic mechanisms to evade T cell-mediated immune surveillance. As a dynamic and reversible modification of the epitranscriptome, m6A modification affects the RNA stability and translation processes, playing an important role in the occurrence and progress of tumors.
However, how m6A modifications participate in regulating the tumor immune microenvironment and regulating T cell-mediated immune surveillance is still unclear.
Recently, a collaborative team led by professor HAN Dali from Beijing Institute of Genomics of the Chinese Academy of Sciences (China National Center for Bioinformation), professor XU Michelle Meng from Tsinghua University and professor YANG Caiguang from Shanghai Institute of Materia Medica reveal that the m6A demethylase FTO could enhance the glycolytic metabolism of tumor cells through epitranscriptomic regulation, thereby inhibiting T cell activation and promoting immune evasion, this paper was published in Cell Metabolism on April 27, 2020.
Researchers have found that knocking down the m6A demethylase FTO in various solid tumors can inhibit tumor growth in mice and increase the proportion of tumor-infiltrating antigen-specific CD8+ T cells.
Further studies have shown that knocking down FTO in tumors leads to faster activation of CD8+ T cells, promoting their antitumor capability.
Through integrated analysis of multidimensional omics sequencing data, including RNA-seq, MeRIP-seq (m6A-seq), ATAC-seq, ChIP-seq, researchers found that FTO can specifically remove the m6A modification on mRNA transcripts of multiple bZIP family transcription factors, thus facilitating their mRNA stability.
These bZIP family transcription factors can promote the expression of glycolytic genes and enhance the glycolytic activity of tumor cells, which in turn inhibits the activation of CD8+ T cells.
Next, the researchers used the small molecule inhibitor Dac51 that targets FTO to further verify the mechanism of FTO in regulating glycolysis metabolism and escaping immune surveillance.
Subsequently, they conducted PD-L1 blockade and Dac51 treatment in tumor-bearing mice model, and found that both can effectively inhibit tumor growth in mice, while the combination of PD-L1 and Dac51 can greatly improve the therapeutic effect.
In addition, the researchers found that Dac51 treatment can also effectively enhance the antitumor immune response of T cells in patient-derived organoid model, suggesting the possibility of Dac51 in clinical applications.
In summary, this study uncovers that RNA epitranscriptome can be operating as an additional layer of genetic regulation for immune evasion, which would facilitate the discovery of a new class of potentially vulnerable epitranscriptomic immunotherapy targets.
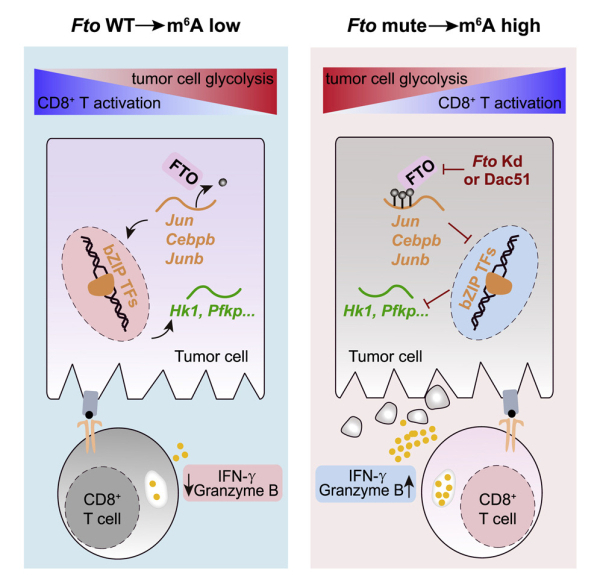
m6A demethylase FTO promotes tumor glycolysis and restricts T cell responses. Treatment with the FTO inhibitor Dac51 increases CD8+ T cell infiltration in tumors and synergizes with anti-PD-L1 blockade (Image by Dali Han's group, Caiguang Yang's group and Meng Michelle Xu's group)
Contact:
Prof. HAN Dali
Email: handl@big.ac.cn