New Mechanism Revealed for Tumor-associated Macrophages to Regulate the Fate of CD8+T Cells
Immune checkpoint blockade therapy is an efficient way to reinvigorate CD8+ T cells, which has demonstrated clinical benefits in multiple types of cancer. However, many patients do not respond to immunotherapies effectively due to the irreversibly dysfunctional state of tumor-infiltrating T cells. Tumor-associated macrophages (TAMs) are the dominant population to induce T cells dysfunction. They are plastic and heterogeneous, and the plasticity could be precisely regulated by dynamic epitranscriptome, coordinately with transcriptional regulation.
As the most abundant modification in eukaryotic mRNA, N6-methyladenosine (m6A) could affect the fate of mRNA by regulating the half-life or translational efficacy of mRNA. Recent studies have pointed out that the m6A abundance and m6A modifiers are dysregulated in cancers. However, whether m6A modification is involved in regulating the function of tumor-infiltrating immune cells and orchestrating an immunosuppressive tumor microenvironment to induce T cells dysfunction is still unclear.
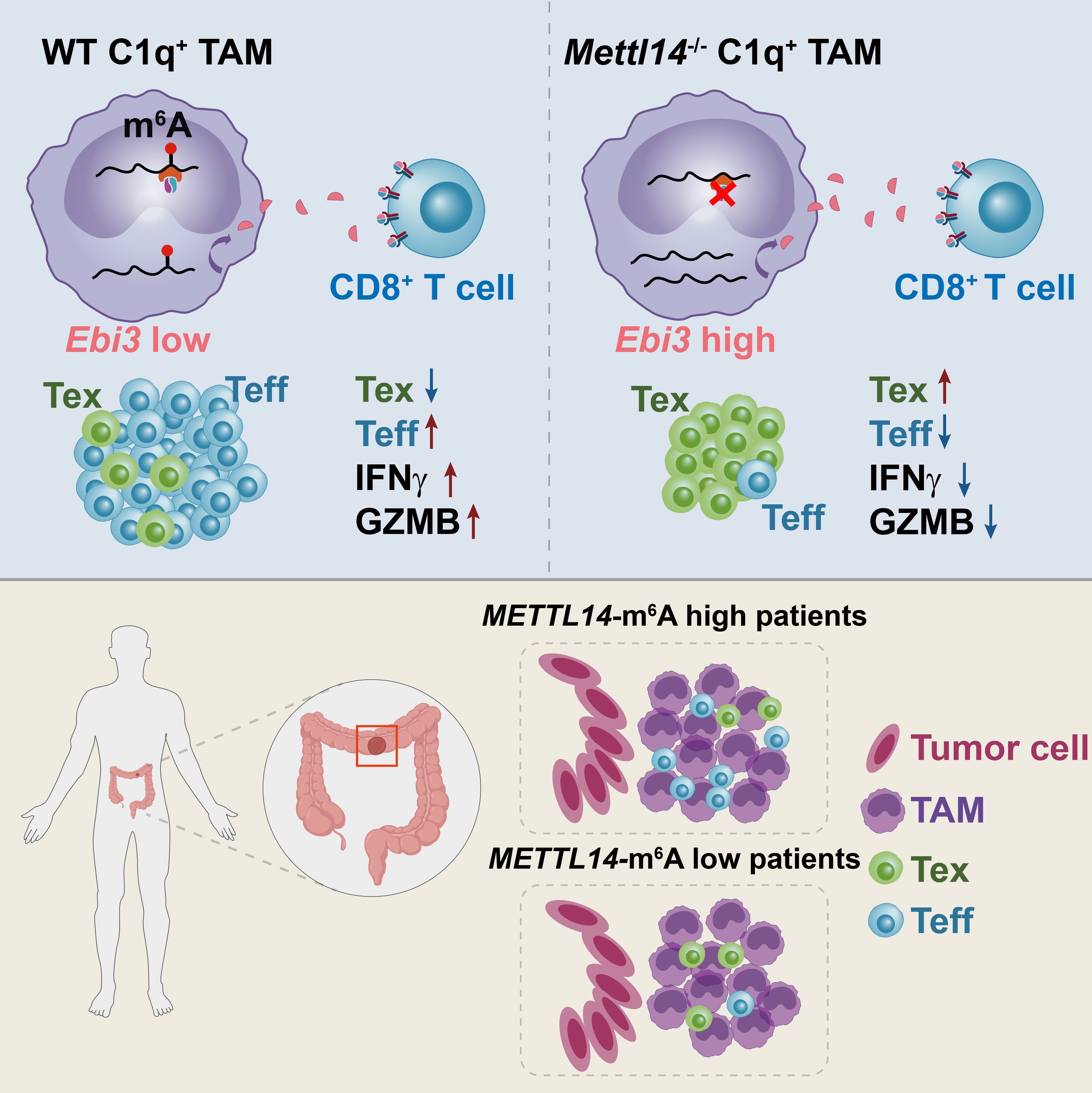
In a study published in Cancer Cell, a collaborative team led by Prof. HAN Dali from Beijing Institute of Genomics of the Chinese Academy of Sciences (China National Center for Bioinformation) and Prof. XU Michelle Meng from Tsinghua University revealed that the loss of m6A methylase METL14 in C1q+ macrophages leads to a decrease in m6A modification and an increase in expression level on Ebi3, which in turn induces the dysfunction of tumor-infiltrating CD8+ T cells.
The researchers revealed that C1q+ TAMs express a set of immunomodulatory ligands to interact with T cells. Notably, m6A methylation-associated genes, such as Mettl14, was highly enriched in C1q+ TAMs.
To investigate the function of m6A modification in macrophages, they specifically depleted Mettl14 in macrophages and found that the capacity to eliminate tumors was impaired and the proportion of CD8+ T cells decreased in Mettl14 deficient mice. Single cell RNA sequencing data demonstrated that Mettl14 deficiency in macrophages abolished the maintenance of effector and progenitor exhausted CD8+ T cell whereas increased the infiltration of dysfunctional transitory CD8+ T cells. Experiment for functional phenotype of tumor-infiltrating CD8+ T cells further validated that cytotoxic cytokine production of CD8+ T cells was impaired in Mettl14 deficient mice.
These data revealed that Mettl14 deficiency in macrophages control the bifurcation between divergent CD8+ T cell fates, dampening the effector CD8+ T cell activation and driving CD8+ T cell dysfunction.
Besides, the researchers found that the m6A abundance of Ebi3 was remarkably decreased and the mRNA and protein level of Ebi3 were markedly upregulated in Mettl14-deficient macrophages through integrated analysis of m6A-seq and RNA-seq data between wild-type and Mettl14-deficient macrophages. Subsequently, they treated tumor-bearing mice with an anti-EBI3 neutralizing antibody and found that T cell effector function was largely rescued in mice with Mettl14-deficient, which further improved the antitumor ability of Mettl14-deficient mice.
These findings supported that the loss of m6A in TAMs induces CD8+ T cell dysfunction by facilitating the accumulation of EBI3. To determine whether these findings can translate to human patients’ tumor samples, the researchers conducted multi-color immunohistochemistry and observed macrophages were close to CD8+ T cells in colon cancer patients.
In addition, they found that METTL14 expression level in stromal cells was positively correlated with the overall m6A level and CD8+ T cells infiltration within tumor. Furthermore, patients with higher m6A levels in stromal cells showed higher effector T cell signatures.
This study revealed a new mechanism that Mettl14 deficiency in macrophages can promote the accumulation of Ebi3, thereby driving CD8+ T cell dysfunction. It also showed that the function of macrophages was regulated by m6A modification at the epitranscriptomic level and emphasized that the functional plasticity of macrophages could be precisely switched through the dynamic epitranscriptome. Blocking the downstream molecules of Mettl14, such as EBI3, should be hopeful to restrict T cell dysfunction and improve the responsiveness of immune checkpoint blockade.
The loss of m6A in tumor-associated macrophages promotes CD8+ T cell dysfunction (Image by HAN Dali's group and XU Michelle Meng's group)
Contact:
Prof. HAN Dali
Email: handl@big.ac.cn