Scientists Build 'Digital Aging Twin' to Track How the Body Wears Down
A large research consortium, the Aging Biomarker Consortium (ABC), has built a comprehensive "digital aging twin" framework. It can predict a person's biological age, track how individual organs age at different rates, and identify a key driver of age-related decline—coagulation factors accumulating in the blood. Published in Cell on May 8, the findings mark a major step from simply describing aging to systematically quantifying it and potentially intervening.
Aging is complex, and measuring it precisely has long been a challenge. Two people of the same chronological age can have vastly different health trajectories, and scientists have struggled to move beyond identifying markers of aging to pinpoint what actually drives it. Led by researchers from the Institute of Zoology,the China National Center for Bioinformation at the Chinese Academy of Sciences, Xuanwu Hospital Capital Medical University, and seven other institutions, the new study tackles this problem head-on by creating a "Digital Aging Twin" framework.
The team built a standardized multicenter cohort called mCAS (multicentric Chinese Aging Standardized), recruiting 2,019 healthy individuals aged 18 to 91 from Beijing, Quzhou, Ningbo, and Nanchang. For each participant, they collected over 240 parameters spanning clinical tests, cognitive and motor function assessments, brain and retinal imaging, gait analysis, and six layers of molecular data, including DNA methylation, RNA transcripts, proteins, metabolites, and gut microbiomes. This dataset, representing more than a billion high-quality data points, serves as the foundation for a three-tiered aging clock system.
The first tier, the core capacity clock, integrates 240 physiological indicators to capture overall functional decline. The second and most powerful tier, the multimodal clock, fuses all six omics layers using a deep learning architecture with attention mechanisms to weigh the importance of different data types. This multimodal clock predicted chronological age with a mean absolute error of just 3.87 years, outperforming all single-layer clocks. The third tier consists of organ-specific clocks for the brain, liver, lung, muscle, blood vessels, and skin, each built from clinical markers, plasma proteins, and imaging features.
One of the most striking findings is that organs do not age in sync. The liver shows a critical aging inflection point around age 40, while the brain's acceleration point comes around age 50. The analysis also identified two major nonlinear waves of aging-related change—one in the 40–50 age range and another between 60 and 70. The later wave is marked by pronounced activation of the blood coagulation cascade, with multiple clotting factors showing sharp age-related increases.
The study then moves from correlation to causation. By analyzing plasma proteomics, liver tissue staining from human donors, and experimental models, the team identified age-driven accumulation of liver-derived coagulation factors—particularly F13B, but also F9 and F10—as a direct driver of vascular and systemic aging. When human aortic endothelial cells were exposed to these factors, they showed clear signs of senescence: increased aging markers, impaired tube formation (a measure of blood vessel health), and heightened inflammation. Injecting F13B into mice accelerated aging across multiple tissues, including the liver, heart, aorta, and kidney, accompanied by immune cell infiltration and inflammatory signals. These results position coagulation factors not merely as passive biomarkers but as actionable drivers of aging.
To make the framework clinically practical, the researchers developed simplified "proxy clocks" using just 100 to 108 plasma proteins. These protein-based proxies accurately recapitulate the predictions of the much more complex core capacity clock and organ clocks, suggesting that a relatively simple blood test might one day provide a comprehensive aging assessment. The study also identified lifestyle factors associated with accelerated versus decelerated aging. Regular fruit intake, appropriate sleep timing, and moderate walking correlated with slower aging, while smoking, insufficient sleep, and high meal frequency correlated with faster aging.
The research is the first proof-of-concept for the X-Age Project (also known as "Mao Die"), a major national initiative led by the Aging Biomarker Consortium to establish a comprehensive aging clock system for the Chinese population. While the current framework is built on cross-sectional data, it is being continuously refined with longitudinal follow-up and expanded populations. Future iterations will also develop lower-cost, more sensitive detection methods. The study acknowledges limitations, including the need for independent validation of coagulation factor inhibitors and the inherent constraints of cross-sectional data, which captures snapshots rather than true trajectories.
Nevertheless, the Digital Aging Twin framework represents a fundamental shift in aging science—from describing what happens to predicting what will happen, and from identifying correlations to pinpointing drivers. Researchers now have a standardized, quantifiable, and interpretable system that can answer not just how old a person is, but how fast they are aging, which organs are leading the decline, and where interventions might be most effectively targeted.
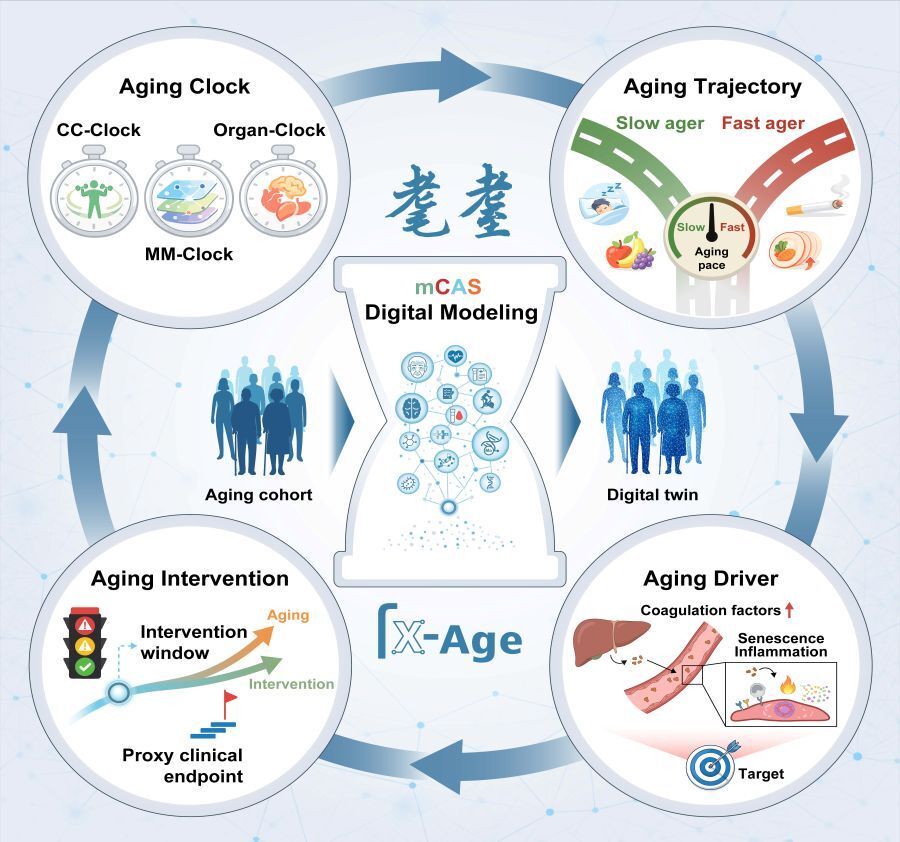
Figure X-Age framework for multidimensional aging assessment and digital modeling