m6A Facilitates Transcription Termination Through Promoting R-loop Formation
R-loops are three-stranded nucleic acid structures with a DNA-RNA heteroduplex and a displaced DNA single strand. It is a widespread genomic feature in organisms from prokaryotes to eukaryotes, as high-throughput mapping studies revealed their prevalent presence on tens of thousands of genomic loci. These loci correspond with important roles that they play in many cellular processes, such as immunoglobulin class switching, mitotic chromosome segregation, various transcription stages and other chromatin regulations.
Chromatin-association of nascent RNA, special DNA structures including G-quadruplex, and negative supercoiling behind elongating RNA polymerase II prompt R-loop formation. Meanwhile, quite a few R-loop dissolution mechanisms have also been identified, including RNase H, several helicases and RNA binding factors. All these major advances highlighted the importance of many layers of regulations for the properties and functions of R-Loop, but all through external elements. It is still unclear whether or not changing the intrinsic property of RNA itself, such as various RNA modifications, could modulate the formation of RNA:DNA hybrids.
A recent cooperative study led by Prof. YANG Yungui and Prof. REN Jie from Beijing Institute of Genomics, Chinese Academy of Sciences and Prof. SUN Qianwen from Tsinghua University presented the first evidence that, indeed, one RNA modification, m6A promotes R-loop formation. Moreover, such modification constitutes a critical step in the dynamic regulation of R-loop at the termination stage of transcription, to prevent the read-through activity and to promote proper release of RNA polymerase II. This work has, for the first time, demonstrated that RNA modification itself acts as a novel cis-regulating element in R-Loop formation and transcription termination, and published in Cell Research on Oct 12.
The team first found that knocking-down the m6A methyltransferase METTL3 significantly reduces R-loop signals by dot-blot and immunofluorescence assays using S9.6 antibody. And this regulation was shown to depend on the methyltransferase activity of METTL3. Using UHPLC-MRM-MS/MS, they showed that m6A modifications are more enriched in R-loops than total chromatin-associated RNA.
Further, using their newly developed ssDRIP-seq method, which provides highly sensitive and strand-specific information of R-loop distribution, they revealed, for the first time, a synergistic role between R-loop formation and METTL3-mediated m6A modification at a genome-wide scale. R-loops were decreased significantly in regions that flank transcription end sites (TES) and harbor m6A sites (m6A+ genes) at the same time. And this reduction can be restored by wild-type METTL3, but not the methyltransferase deficient one.
In order to explore the biological function of m6A’s regulation on R-loops, the team measured the precise location of RNA polymerase II using mNET-seq, and observed a significant readthrough activity in METTL3 deficient cells at transcription termination regions of genes with m6A modification. Using a reporter system testing readthrough activities downstream of R-loop-terminated transcription, they validated that METTL3 knockdown impairs transcription termination. Thus, METTL3-mediated m6A modification on R-loop acts as a cis-regulating element in transcription termination.
These findings revealed a novel co-transcriptional regulatory role of RNA m6A modification in promoting R-loop formation for proper and efficient transcription termination. It will provide theoretical and technical support for further investigation of the underlying mechanism and extended biological functions.
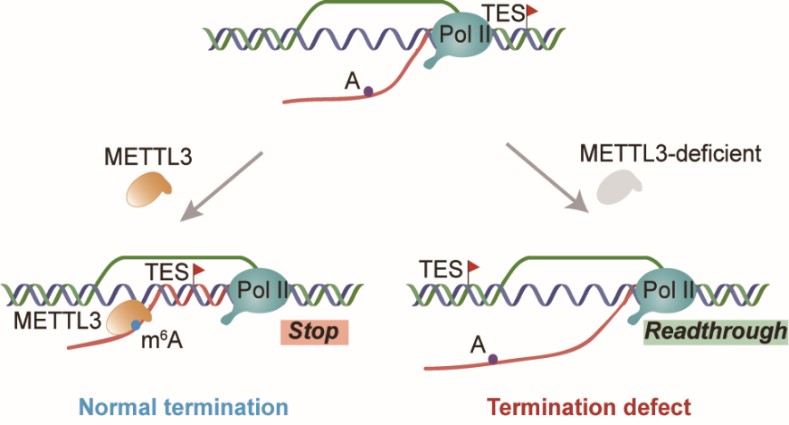
m6A promotes R-loop formation for efficient transcription termination (Image by YANG Yungui's group)
Contact:
Dr. YANG Yungui
Email: ygyang@big.ac.cn
Dr. REN Jie
Email: renjie@big.ac.cn