Immune Evasion and ACE2 Binding Affinity Contribute to SARS-CoV-2 Evolution
Since the discovery of the SARS-CoV-2 virus at the end of 2019, the virus has been continuously mutating, giving rise to a series of variants, including Delta and Omicron. Many functional mutations that can affect the virus's transmissibility and immune evasion have been observed in the genomes of these variants. Recent studies have found that the occurrence of some mutations on the S gene was associated with immune pressure, but the overall trajectory of SARS-CoV-2 evolution and the contributions of immune pressure to virus evolution are still not well understood.
Prof. LI Mingkun’s team from the Beijing Institute of Genomics, Chinese Academy of Sciences (China National Center for Bioinformation), in collaboration with teams from Peking University and the Changping Laboratory, analyzed two potential evolutionary driving forces of SARS-CoV-2, namely antibody immune evasion and ACE2 binding affinity, and reveals their contributions to virus evolution during the prevalence of different SARS-CoV-2 variants.
This study analyzed 6 million SARS-CoV-2 genomes collected from 2019 to 2022, and found that, except for the significant increase in the number of mutations introduced by the Alpha and Omicron variants, the evolutionary rate of the SARS-CoV-2 genome remained relatively stable. However, newly acquired mutations gradually became concentrated in the receptor-binding domain (RBD) over time, leading to an accelerated evolution rate in this region. Meanwhile, lineage-specific mutation patterns were observed in the RBD region, indicating a tendency for variant with similar RBD sequences to exhibit the same mutations. Notably, this characteristic was not observed in genomic regions outside the RBD region.
The study further utilized deep mutation screening data to characterize the immune evasion capabilities of different mutations in the RBD region. Significant differences in the types of antibodies escaped by mutations in different viral lineages were observed, and the incidence of immune escape mutation was positively correlated with the immune pressure exerted by antibodies on different antigenic epitopes. This correlation strengthened over time, with a Pearson correlation coefficient of 0.8 observed in the BA.5 variant, suggesting that immune pressure resulting from vaccination and natural infection in the population may be the main driving force behind recent SARS-CoV-2 evolution.
Furthermore, the study found that mutations occurring in the early stages of the SARS-CoV-2 pandemic (including the early stage after the emergence of the Omicron variant) had higher ACE2 binding affinity, while later mutations exhibited higher immune evasion capabilities. The study quantified the effects of these two factors on the incidence of the mutations through multiple linear regression analysis and found that both immune evasion capability and ACE2 binding affinity were significantly correlated with the mutation incidence. Their contributions to virus evolution vary at different stages of the pandemic. Further analysis revealed a stronger correlation between immune evasion capability and mutation occurrence, while ACE2 binding affinity was more closely associated with the virus's adaptability in the population (quantified by RoHo).
In conclusion, this study provides a systematic analysis of the evolution of SARS-CoV-2 from 2019 to 2022 and reveals that immune evasion and ACE2 binding affinity are both contributing factors to virus evolution, with their relative importance depending on changes in the population's immune background. The study deepens our understanding of the evolution of the SARS-CoV-2 virus and provides a foundation for predicting its future evolution. It also offers new insights and methods for studying the evolutionary characteristics of other viruses.
This study was published in Nature Ecology & Evolution entitled "Immune evasion and ACE2 binding affinity contribute to SARS-CoV-2 evolution".
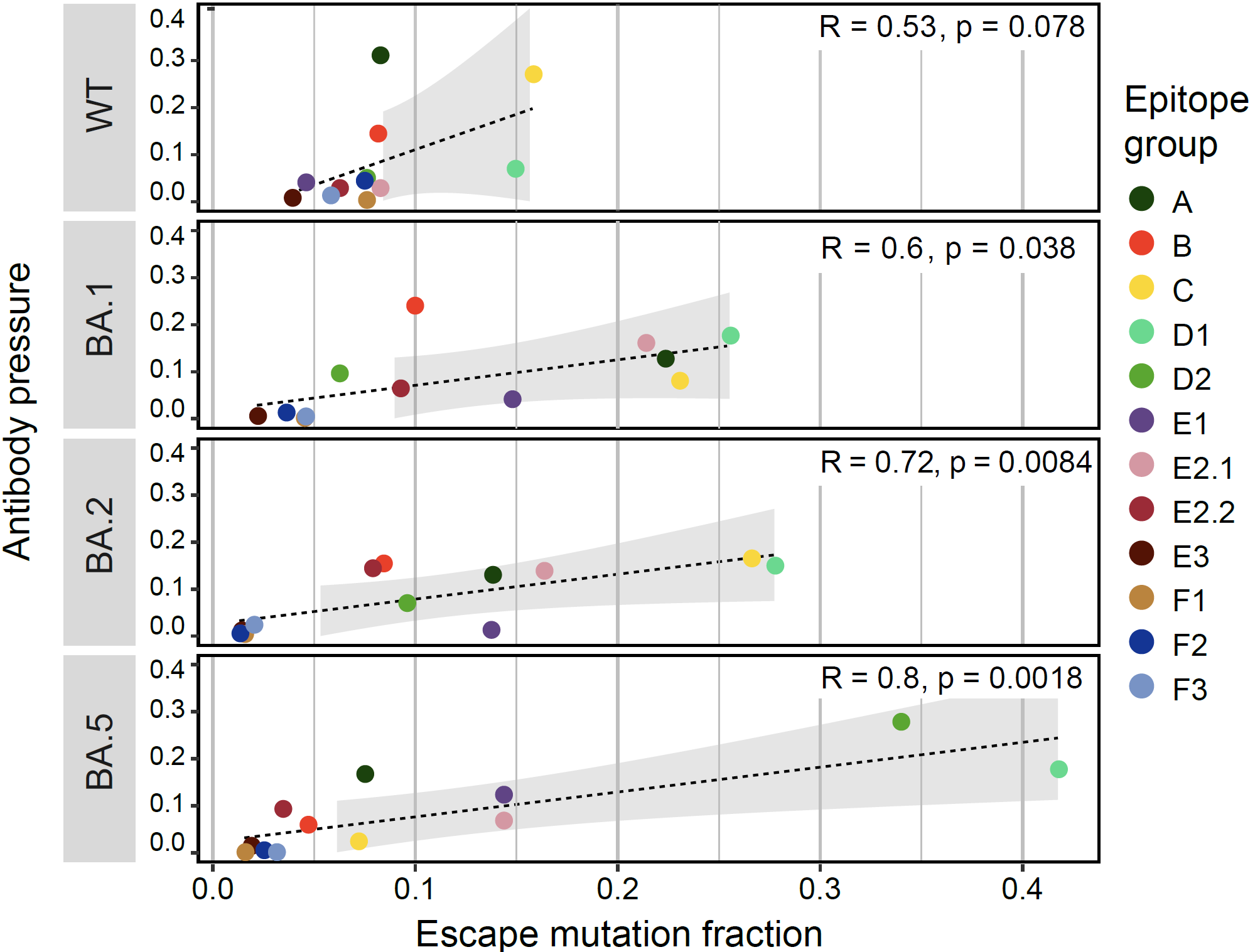
The correlation between the prevalence of escape mutations and antibody pressure in 12 epitope regions.
Contact:
Prof.LI Mingkun
Email: limk@big.ac.cn